Rxmedonline.com: Your Convenient Pharmacy Portal - Page 2
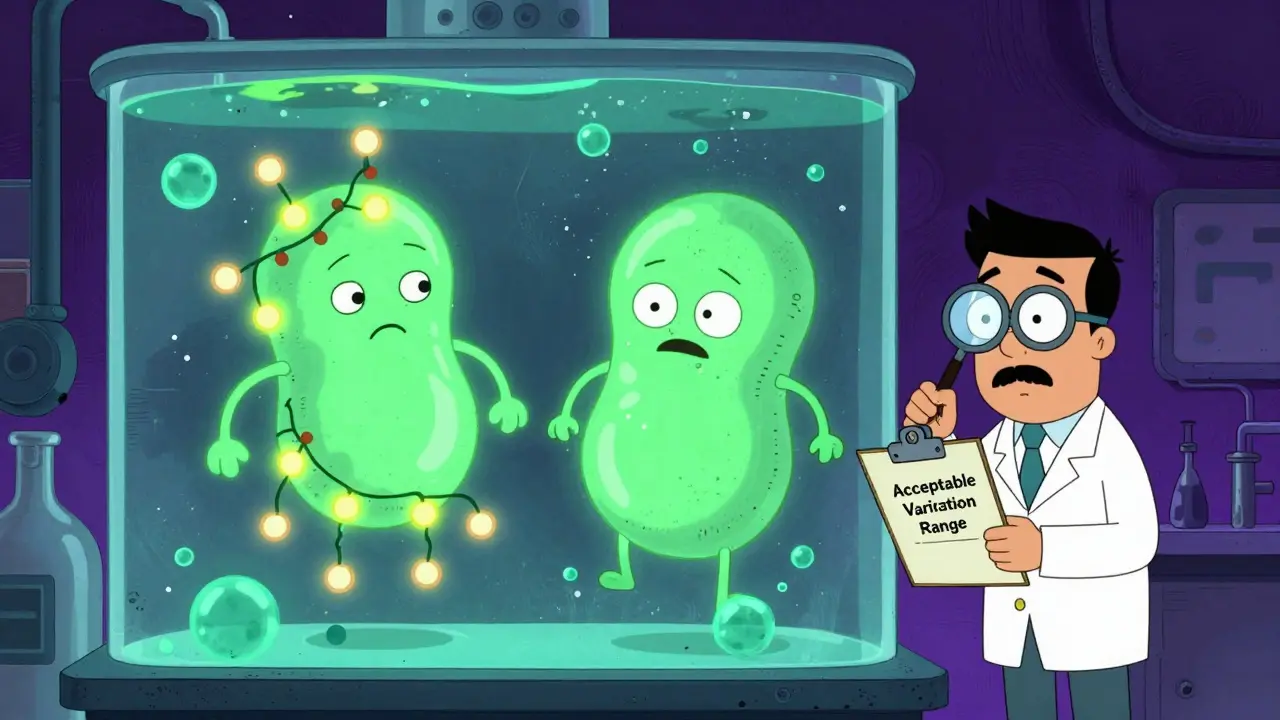
Lot-to-Lot Variability in Biologics and Biosimilars: Why Differences Between Batches Are Normal and Safe
Lot-to-lot variability is a natural part of biologics and biosimilars, not a defect. Learn why batch differences are expected, how regulators ensure safety, and how this enables affordable access to life-saving treatments.
MAO Inhibitors: Dangerous Interactions with Common Medications
MAO inhibitors can save lives in treatment-resistant depression-but they carry deadly risks when mixed with common medications, supplements, or foods. Learn the dangerous interactions that can trigger hypertensive crisis or serotonin syndrome.
Finding Generic Combinations: When Components Don't Match Brand Formulations
Generic combination drugs save money but can differ in inactive ingredients from brand versions-potentially affecting absorption, side effects, and effectiveness. Learn which patients are at risk and how to spot dangerous formulation changes.
Navigating Medication Safety in Hospitals and Clinics: Key Practices to Prevent Errors
Medication errors kill thousands each year in U.S. hospitals. Learn how ISMP's targeted safety practices, barcode systems, and patient involvement are preventing deadly mistakes-and why so many hospitals still aren't following through.
Allergy and Cold Medications: Combination Safety and Interactions
Combination cold and allergy medications may seem convenient, but mixing ingredients like acetaminophen, phenylephrine, and dextromethorphan can lead to dangerous interactions, liver damage, or serotonin syndrome. Learn what's really in your medicine and how to avoid hidden risks.
Chest Pain Evaluation: When to Go to the Emergency Department
Chest pain can signal a heart attack-or something less serious. Learn the warning signs that require an emergency room visit, what tests doctors use, and why calling 9-1-1 beats driving yourself. Based on 2021 AHA/ACC guidelines.
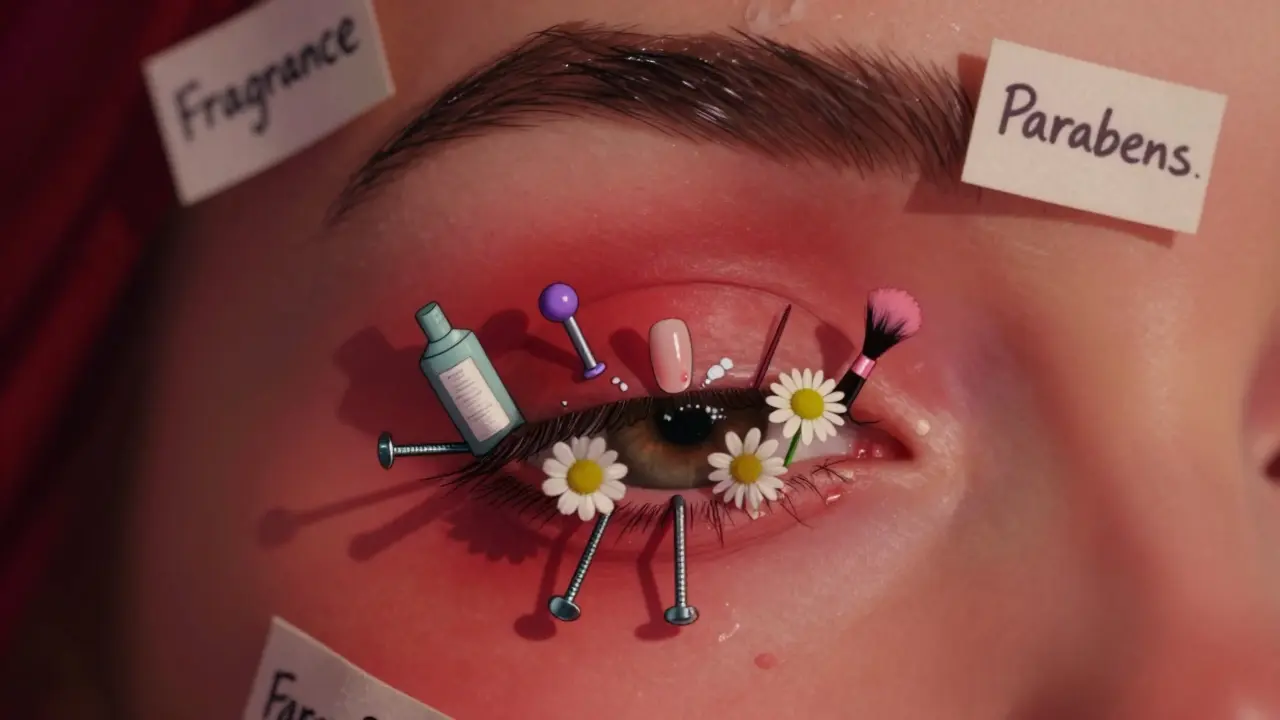
Eyelid Dermatitis: How Cosmetic Allergens Cause It and What Actually Works
Eyelid dermatitis is often caused by hidden allergens in cosmetics, nail polish, and hair products. Learn the top triggers, why patch testing is essential, and how to treat it safely without damaging your eyes.
MedWatch System Explained: How FDA Tracks Drug and Device Safety
MedWatch is the FDA's system for tracking adverse events from drugs, devices, and other medical products. Learn how reports from patients and providers help the FDA detect safety risks and protect public health.
Pharmacist Role with Biosimilars: Counseling and Substitution
Pharmacists play a critical role in biosimilar adoption by counseling patients, navigating complex substitution laws, and ensuring traceability. Unlike generics, biosimilars require specialized knowledge - and their success depends on pharmacist-led education and policy alignment.
Liability and Indemnification in Generic Transactions Explained
Indemnification in commercial transactions is a legal mechanism that shifts financial risk between parties. Learn how it works, what clauses to demand, and how to avoid costly mistakes in contracts.